
NHS
World’s first lung cancer vaccine begins trials: here’s what we know so far
2 years ago

A groundbreaking clinical trial testing the safety of a new lung cancer vaccine, BNT116, has commenced across England and Wales, marking a significant milestone in cancer treatment.
The first UK patient received the new lung cancer vaccine on Tuesday at the National Institute for Health Research UCLH Clinical Research Facility in London, with another patient set for trail at the Clatterbridge Centre in Wirral.
Here’s what we know so far:
But what is the vaccine and how does it work?
The vaccine, known as BNT116, utilises messenger RNA (mRNA), the same technology used in the Covid-19 vaccines.
The jab works by presenting the immune system with tumour markers from non-small cell lung cancer (NSCLC). This primes the body to fight the cancer cells that are expressing these markets while leaving healthy cells untouched.
How is it administered?
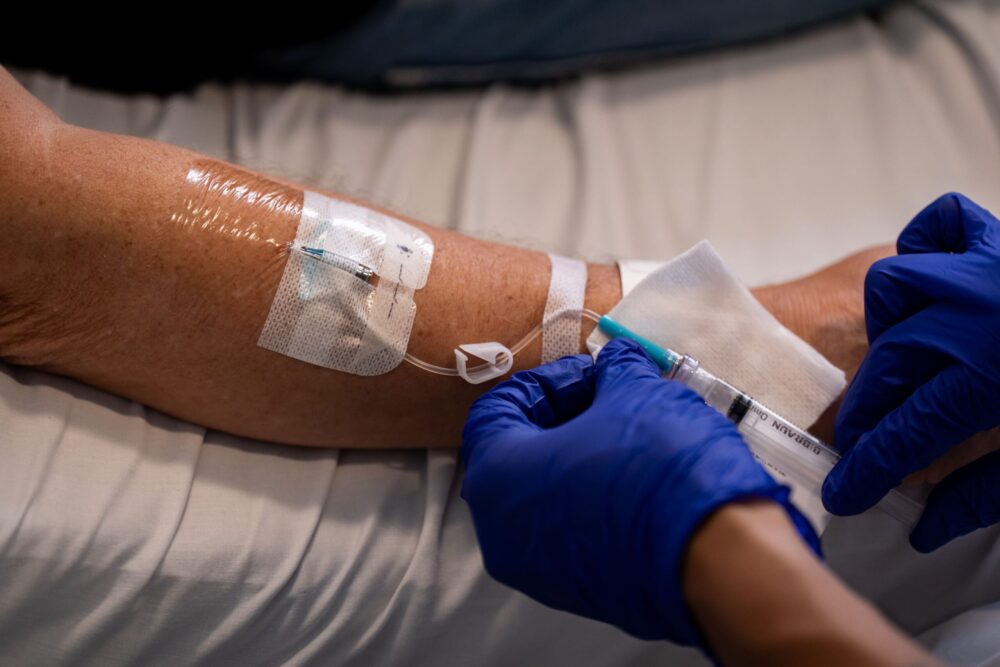
This trial will aim to recruit people with NSCLC – from early-stage before surgery or radiotherapy, to late-stage disease or recurrent cancer – to have the jab alongside standard immunotherapy.
The first patient to have the vaccine – Janusz Racz, 67, from London – had six consecutive injections given five minutes apart over a half hour period.
Each jab contained different RNA strands. He will get the vaccine every week for six consecutive weeks and then every three weeks for a year and then every three weeks for a total of 54 weeks.
What are the potential outcomes for lung cancer patients who have the jab?
Professor Siow Ming Lee, a consultant medical oncologist at University College London Hospitals NHS Foundation Trust (UCLH), which is leading the trial in the UK, said he hopes the vaccine will prevent lung cancer from coming back.
Immunotherapy has made “big progress”, according to Prof Lee, but still does not treat all lung cancer patients successfully.
He describes the jab as “another additional immune approach attack” and said it could be an “extra boost” to improve survival rates for people with the disease.
What is the scale of the trial?

The phase one clinical trial will take place at 34 sites across seven countries.
Six are located across England and Wales, with the National Institute for Health Research UCLH Clinical Research Facility leading the trial in the UK.
Other hospital sites are Cambridge University Hospitals NHS Foundation Trust, Velindre University NHS Trust in Cardiff, The Clatterbridge Cancer Centre NHS Foundation Trust on the Wirral, Guy’s and St Thomas’ NHS Foundation Trust in London, and The Newcastle Upon Tyne Hospitals NHS Foundation Trust.
Overall, is it hoped about 130 lung cancer patients will be recruited for the study, with 20 based in the UK.
What does the trial hope to achieve and what could happen next?
As a phase one study, this trial will determine the safety of the BNT116 vaccine.
When a phase one trial is successful, researchers usually move on to phase two, which will look at effectiveness and usually includes a larger number of patients. A phase three trial is larger still and usually compares a new treatment to standard treatment.
Prof Lee said he hopes this vaccine eventually “becomes standard of care worldwide and save lots of lung cancer patients”.
Are there vaccines for other types of cancer?
Yes. In April, a final phase three trial involving a personalised mRNA jab for melanoma was launched at UCLH.
The vaccine is custom-built for each patient in just a few weeks and works by telling the body to hunt down cancer cells and prevent the disease from coming back.
A phase two trial, involving pharma firms Moderna and MSD, found the treatment dramatically reduced the risk of the cancer returning in melanoma patients.
In June, it also emerged the first patient in England had received an mRNA vaccine for bowel cancer, which is being developed by BioNTech and Genentech. The trial at Queen Elizabeth Hospital Birmingham forms part of NHS England’s Cancer Vaccine Launch Pad.
What do jabs like these mean for cancer care and what could the future hold?
Prof Lee described vaccines like this as “the next big phase of cancer treatment”.
BNT116 is made by BioNTech, which signed an agreement with the Government in July 2023 to provide up to 10,000 patients with precision cancer immunotherapies by 2030.
NHS England’s Cancer Vaccine Launch Pad is also working to fast-track patients to get vaccines at the earliest opportunity.











 Subscribe
Subscribe Follow Us
Follow Us Follow Us
Follow Us Follow Us
Follow Us Follow Us
Follow Us











