
Coronavirus
Key questions about the Pfizer vaccine announcement and what it means for you
5 years ago
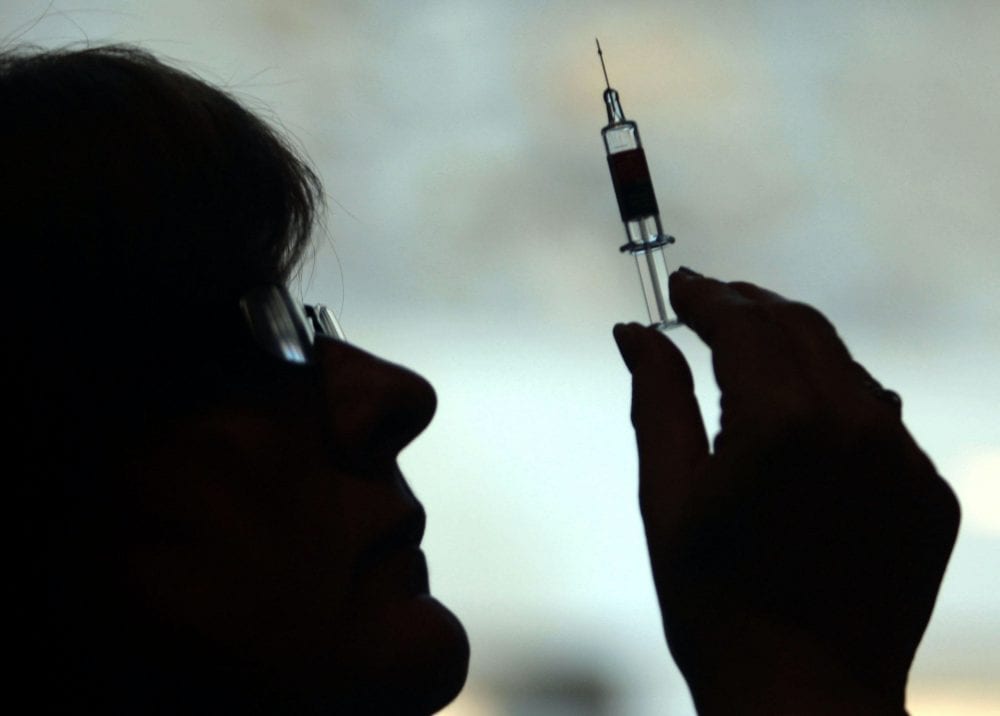
There is hope of a new coronavirus vaccine with data suggesting the vaccine created by Pfizer and BioNTech is effective, and there are more than 200 other vaccines currently being tested around the world.
The pharmaceutical giant Pfizer, working with German biotech company BioNTech, has released interim analysis suggesting their vaccine is more than 90% effective in preventing Covid-19.
A coronavirus vaccine could start being distributed by Christmas after a jab developed by pharmaceutical giant Pfizer cleared a “significant hurdle”.
Prime Minister Boris Johnson said initial results suggested the vaccine was 90% effective at protecting people from Covid-19 but warned these were “very, very early days”.
The announcement from Pfizer and its vaccine partner BioNTech was hailed by scientists as a significant breakthrough in the fight against coronavirus, while stock markets rallied on the news with the FTSE 100 jumping more than 5.5%.
Dr Richard Vautrey, chairman of the British Medical Association’s GP committee in England said practices would “stand ready” to deliver the vaccine, with clinics potentially running from 8am-8pm, seven days a week.
Deputy chief medical officer Professor Jonathan Van-Tam told a Downing Street press conference he was “hopeful” there would be “some vaccine by Christmas”.
He said: “Frankly, we’re in the middle of the second wave, and I don’t see the vaccine making any difference for the wave we are now in.
“I’m hopeful that it may prevent future waves, but this one we have to battle through to the end without a vaccine.
“This is a very important scientific breakthrough. I am certain of that.
“I am hopeful because of all that, but not yet certain that we could begin to see some vaccine by Christmas.”

Here is everything you need to know about the coronavirus vaccine race.
What progress is being made with Covid-19 vaccines?
There are currently more than 200 coronavirus vaccine candidates being tested around the world.
About 12 Covid-19 vaccines around the world are currently in the final stages of testing, but the one from German biotech firm BioNtech and US pharmaceutical company Pfizer is the first to report any results.
There are two frontrunners in the Covid-19 vaccine race – the one from Pfizer, called BNT162b2, and another being developed by the University of Oxford and AstraZeneca, which is also in phase three clinical trials.
Other potential vaccines in phase three trials include ones by US drugs firm Moderna and biotech company Novavax.
Is the Pfizer vaccine announcement big news?
Yes. These are interim findings and studies will continue but analysis shows that the Pfizer vaccine can prevent more than 90% of people from getting Covid-19.
The vaccine has been tested on 43,500 people in six countries and no safety concerns have been raised.
Pfizer plans to apply to the US regulator the Food and Drug Administration (FDA) for emergency approval to use the vaccine by the end of the month.
The analysis was carried out after 94 confirmed cases of Covid-19 were found among those taking part in the trial.
What type of vaccine is this?
The jab is known as a messenger RNA (mRNA) vaccine.
Conventional vaccines are produced using weakened forms of the virus, but mRNAs use only the virus’s genetic code.
An mRNA vaccine is injected into the body where it enters cells and tells them to create antigens.
These antigens are recognised by the immune system and prepare it to fight coronavirus.
What are the advantages of this type of vaccine?
No actual virus is needed to create an mRNA vaccine. This means the rate at which the vaccine can be produced is dramatically accelerated.
As a result, mRNA vaccines have been hailed as potentially offering a rapid solution to new outbreaks of infectious diseases.
In theory, they can also be modified reasonably quickly if, for example, a virus develops mutations and begins to change.
mRNA vaccines are also cheaper to produce than traditional vaccines. But both will play an important role in tackling Covid-19.

Are they safe?
All vaccines undergo rigorous testing and have oversight from experienced regulators.
Some believe mRNA vaccines are safer for the patient as they do not rely on any element of the virus being injected into the body.
mRNA vaccines have been tried and tested in the lab and on animals but the coronavirus vaccine will be the first one licensed for use in humans.
The human trials of mRNA vaccines – involving tens of thousands of people – have been going on since early 2020 to show whether it is safe and effective.
Pfizer will continue to collect safety and long-term outcomes data from participants for two years.
Do we have enough doses to vaccinate the UK population?
The UK has secured 40 million doses of the Pfizer/BioNTech vaccine – the first agreement the firms signed with any government.
People will need two doses, meaning not enough shots have been secured for the entire UK population.
However, it is likely other vaccines will announce results from their clinical trials shortly.
Who will be vaccinated first?
The Joint Committee on Vaccination and Immunisation (JCVI) has examined data on who suffers the worst outcomes from coronavirus and who is at highest risk of death.
Its interim guidance says the order of priority should be:
– Older adults in a care home and care home workers
– All those aged 80 and over and health and social care workers, though they may move up the list
– Anyone 75 and over
– People aged 70 and over
– All those aged 65 and over
– High-risk adults under 65
– Moderate-risk adults under 65
– All those aged 60 and over
– All those 55 and over
– All those aged 50 and over
– The rest of the population, with priority yet to be determined.
When can we expect results from the Oxford vaccine?
Over 20,000 volunteers are now participating in trials for the Oxford vaccine, which are taking place in countries including the UK, South Africa, Brazil and Kenya.
Professor Andrew Pollard, head of Oxford’s vaccine trial team, said he is optimistic data on safety and efficacy of their vaccine will be available by the end of the year.
Appearing before the Commons Science and Technology Committee, he said there is a “small chance” of a vaccine being made available by Christmas.
The Oxford vaccine, called ChAdOx1 nCoV-19, uses a weakened version of a common cold virus (adenovirus) which causes infections in chimpanzees.
– What other trials are ongoing in the UK?
Aside from the Oxford vaccine, a coronavirus jab is being developed by Imperial College London.
The Imperial vaccine is in phase one of clinical testing, where doses are being given to a small group of people to determine whether it is safe and to learn more about the immune response it provokes.
Professor Robin Shattock, who is leading Imperial College London’s Covid-19 vaccine effort, said data on its efficacy will be available in the middle of next year.
Pharmaceutical companies Sanofi and GlaxoSmithKline have also teamed up with the hope of making a Covid-19 vaccine available by the middle of next year.
The Sanofi/GSK candidate is in the phase two stage, where the vaccine is being given to hundreds of people so scientists can learn more about its safety and correct dosage, and plan to begin phase three by the end of the year.
Does the UK have access to any of these potential vaccines?
The UK has secured 40 million doses of the Pfizer/BioNTech vaccine, the first agreement the firms signed with any government.
In August, the Government announced the UK has secured access to six Covid-19 vaccine candidates in development, representing 340 million doses.
The deals cover four different types of vaccines – adenoviral vaccines, mRNA vaccines, inactivated whole virus vaccines and protein adjuvant vaccines.
Adenoviral vaccines are weakened versions of adenoviruses, while mRNA candidates use the virus’s genetic code, as with the Pfizer/BioNTech jab.
Inactivated whole virus vaccines, on the other hand, contain whole bacteria or viruses which have been killed, while protein adjuvant jabs are those where an adjuvant is added to enhance the immune response.
When could a vaccine become available in the UK?
A vaccine usually takes years, often decades, to develop, but scientists working on potential coronavirus jabs are hoping to achieve the same amount of work in a few months.
Pfizer and BioNTech plan to apply to the US Food and Drug Administration – the US medicines regulator – by the end of this month for emergency approval to use the vaccine.
Downing Street welcomed the results as “promising” and said the UK will have procured 10 million doses by the end of the year to be given out if it is approved.
NHS England chief executive Sir Simon Stevens said the “expectation” is that any vaccination programme would begin in the new year – pending positive results from clinical trials.
Kate Bingham, chairwoman of the UK vaccine taskforce, said she has 50% confidence that by Easter or early summer next year, all vulnerable people in the country will have a vaccine.








 Subscribe
Subscribe Follow Us
Follow Us Follow Us
Follow Us Follow Us
Follow Us Follow Us
Follow Us Follow Us
Follow Us











